4 results
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization – ERRATUM
- Sarah E. Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson, Jinal Makhija, Mary Carl Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary K. Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 07 October 2022, e166
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization
- Sarah Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson Jinal, Makhija, Raheeb Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Joseph Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s78-s79
-
- Article
-
- You have access Access
- Open access
- Export citation
-
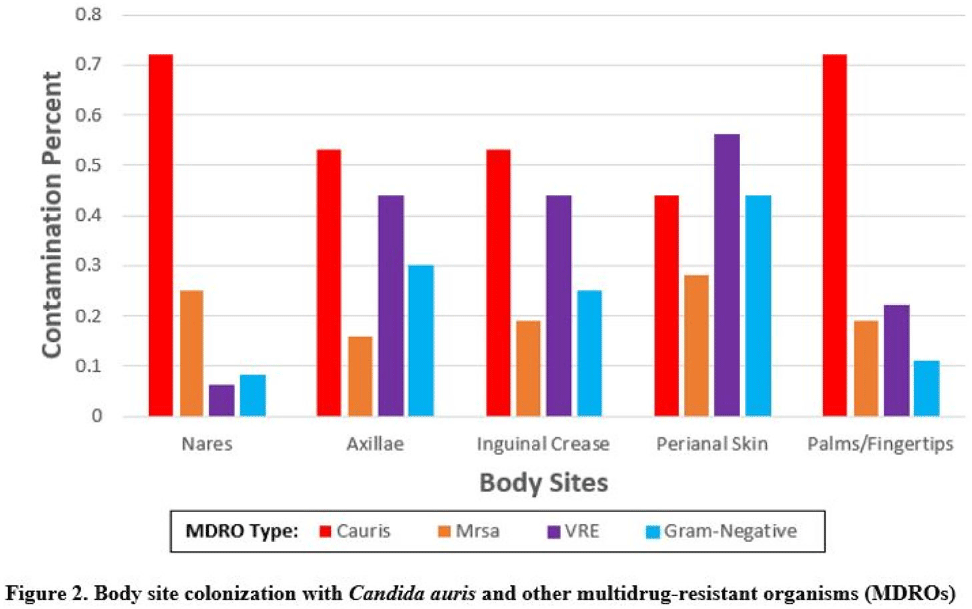
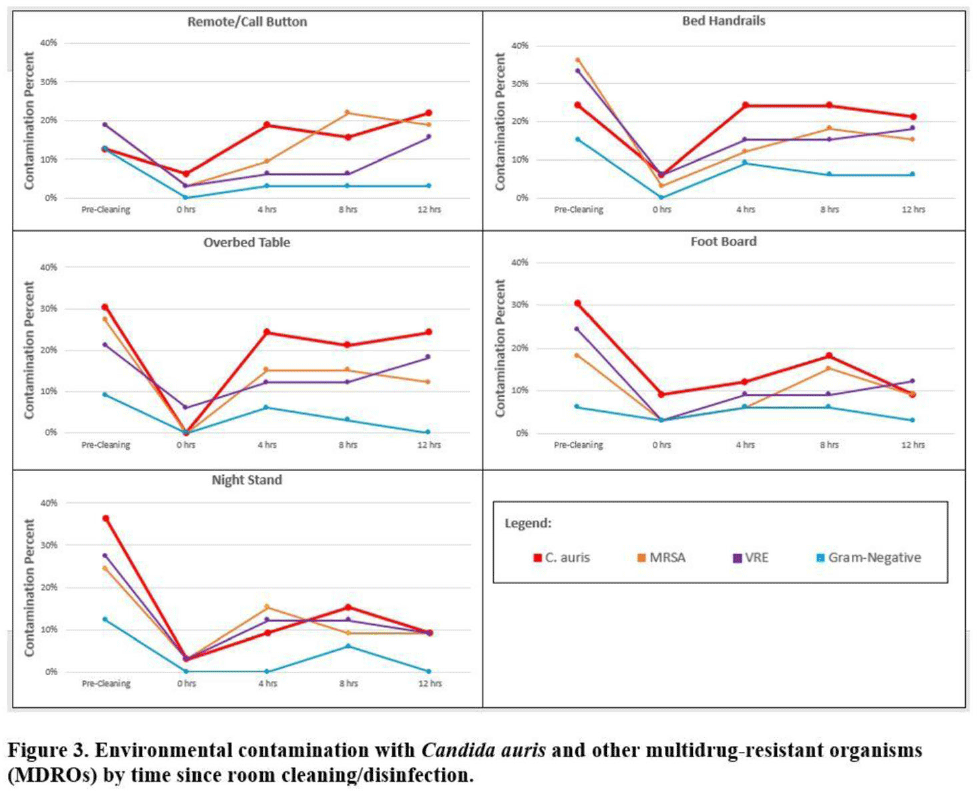
Background:Candida auris is an emerging multidrug-resistant yeast that is transmitted in healthcare facilities and is associated with substantial morbidity and mortality. Environmental contamination is suspected to play an important role in transmission but additional information is needed to inform environmental cleaning recommendations to prevent spread. Methods: We conducted a multiregional (Chicago, IL; Irvine, CA) prospective study of environmental contamination associated with C. auris colonization of patients and residents of 4 long-term care facilities and 1 acute-care hospital. Participants were identified by screening or clinical cultures. Samples were collected from participants’ body sites (eg, nares, axillae, inguinal creases, palms and fingertips, and perianal skin) and their environment before room cleaning. Daily room cleaning and disinfection by facility environmental service workers was followed by targeted cleaning of high-touch surfaces by research staff using hydrogen peroxide wipes (see EPA-approved product for C. auris, List P). Samples were collected immediately after cleaning from high-touch surfaces and repeated at 4-hour intervals up to 12 hours. A pilot phase (n = 12 patients) was conducted to identify the value of testing specific high-touch surfaces to assess environmental contamination. High-yield surfaces were included in the full evaluation phase (n = 20 patients) (Fig. 1). Samples were submitted for semiquantitative culture of C. auris and other multidrug-resistant organisms (MDROs) including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), extended-spectrum β-lactamase–producing Enterobacterales (ESBLs), and carbapenem-resistant Enterobacterales (CRE). Times to room surface contamination with C. auris and other MDROs after effective cleaning were analyzed. Results:Candida auris colonization was most frequently detected in the nares (72%) and palms and fingertips (72%). Cocolonization of body sites with other MDROs was common (Fig. 2). Surfaces located close to the patient were commonly recontaminated with C. auris by 4 hours after cleaning, including the overbed table (24%), bed handrail (24%), and TV remote or call button (19%). Environmental cocontamination was more common with resistant gram-positive organisms (MRSA and, VRE) than resistant gram-negative organisms (Fig. 3). C. auris was rarely detected on surfaces located outside a patient’s room (1 of 120 swabs; <1%). Conclusions: Environmental surfaces near C. auris–colonized patients were rapidly recontaminated after cleaning and disinfection. Cocolonization of skin and environment with other MDROs was common, with resistant gram-positive organisms predominating over gram-negative organisms on environmental surfaces. Limitations include lack of organism sequencing or typing to confirm environmental contamination was from the room resident. Rapid recontamination of environmental surfaces after manual cleaning and disinfection suggests that alternate mitigation strategies should be evaluated.
Funding: None
Disclosures: None
Regional Impact of a CRE Intervention Targeting High Risk Postacute Care Facilities (Chicago PROTECT)
- Michael Lin, Mary Carl Froilan, Jinal Makhija, Ellen Benson, Sarah Bartsch, Pamela B. Bell, Stephanie Black, Deborah Burdsall, Michelle Ealy, Anthony Fiore, Sharon Foy, Mabel Frias, Alice Han, David Hines, Olufemi Jegede, John Jernigan, Sarah K. Kemble, Mary Alice Lavin, Bruce Lee, George Markovski, Massimo Pacilli, Sujan Reddy, Erica Runningdeer, Michael Schoeny, Mitali Shah, Rachel Slayton, Elizabeth Soda, Nimalie Stone, Angela S. Tang, Karen Trimberger, Marion Tseng, Yingxu Xiang, Robert Weinstein, William Trick, Mary Hayden
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s48-s49
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Carbapenem-resistant Enterobacteriaceae (CRE) are endemic in the Chicago region. We assessed the regional impact of a CRE control intervention targeting high-prevalence facilities; that is, long-term acute-care hospitals (LTACHs) and ventilator-capable skilled nursing facilities (vSNFs). Methods: In July 2017, an academic–public health partnership launched a regional CRE prevention bundle: (1) identifying patient CRE status by querying Illinois’ XDRO registry and periodic point-prevalence surveys reported to public health, (2) cohorting or private rooms with contact precautions for CRE patients, (3) combining hand hygiene adherence, monitoring with general infection control education, and guidance by project coordinators and public health, and (4) daily chlorhexidine gluconate (CHG) bathing. Informed by epidemiology and modeling, we targeted LTACHs and vSNFs in a 13-mile radius from the coordinating center. Illinois mandates CRE reporting to the XDRO registry, which can also be manually queried or generate automated alerts to facilitate interfacility communication. The regional intervention promoted increased automation of alerts to hospitals. The prespecified primary outcome was incident clinical CRE culture reported to the XDRO registry in Cook County by month, analyzed by segmented regression modeling. A secondary outcome was colonization prevalence measured by serial point-prevalence surveys for carbapenemase-producing organism colonization in LTACHs and vSNFs. Results: All eligible LTACHs (n = 6) and vSNFs (n = 9) participated in the intervention. One vSNF declined CHG bathing. vSNFs that implemented CHG bathing typically bathed residents 2–3 times per week instead of daily. Overall, there were significant gaps in infection control practices, especially in vSNFs. Also, 75 Illinois hospitals adopted automated alerts (56 during the intervention period). Mean CRE incidence in Cook County decreased from 59.0 cases per month during baseline to 40.6 cases per month during intervention (P < .001). In a segmented regression model, there was an average reduction of 10.56 cases per month during the 24-month intervention period (P = .02) (Fig. 1), and an estimated 253 incident CRE cases were averted. Mean CRE incidence also decreased among the stratum of vSNF/LTACH intervention facilities (P = .03). However, evidence of ongoing CRE transmission, particularly in vSNFs, persisted, and CRE colonization prevalence remained high at intervention facilities (Table 1). Conclusions: A resource-intensive public health regional CRE intervention was implemented that included enhanced interfacility communication and targeted infection prevention. There was a significant decline in incident CRE clinical cases in Cook County, despite high persistent CRE colonization prevalence in intervention facilities. vSNFs, where understaffing or underresourcing were common and lengths of stay range from months to years, had a major prevalence challenge, underscoring the need for aggressive infection control improvements in these facilities.
Funding: The Centers for Disease Control and Prevention (SHEPheRD Contract No. 200-2011-42037)
Disclosures: M.Y.L. has received research support in the form of contributed product from OpGen and Sage Products (now part of Stryker Corporation), and has received an investigator-initiated grant from CareFusion Foundation (now part of BD).
9 - Hyperhidrosis
-
- By DeeAnna Glaser, Department of Dermatology, Saint Louis University School of Medicine, St Louis, MO, USA, Stephanie Benson, University of Colorado, School of Medicine, Denver
- Edited by Anthony B. Ward, Michael P. Barnes
-
- Book:
- Clinical Uses of Botulinum Toxins
- Published online:
- 02 December 2009
- Print publication:
- 22 March 2007, pp 175-200
-
- Chapter
- Export citation
-
Summary
Introduction
Sweating is a normal physiological response to increasing body temperature and is an important mechanism in releasing heat produced from endogenous and exogenous sources. The heat regulatory center is located in the brain within the hypothalamus, particularly involving the pre-optic and anterior nuclei. These sections of the brain monitor core body temperature through the blood stream, and the body's response to increasing temperature is to release heat through sweat.
Sweating is controlled by the sympathetic nervous system through its preganglionic and postganglionic pathways. Nerve fibers exit the pre-optic or anterior nuclei and descend ipsilaterally through the spinal cord until they reach the intermediolateral column, where they leave the cord and enter the sympathetic chain. The postganglionic fibers leave the chain and join the peripheral nerves until they reach the sweat gland. The neurotransmitter for the sympathetic nervous system is generally norepinephrine; however, the neurotransmitter mainly involved in the sweating process is acetylcholine. Other chemical mediators found in the neuromuscular junction during sweating include vasoactive intestinal peptide, atrial natriuretic peptide, galanin, and calcitonin gene related peptide.
Three glands are important in the creation of sweat: the eccrine, apo-eccrine, and apocrine glands. They are located superiorly in the subcutaneous fat at the junction of the dermis and subcutaneous fat. Between two and four million eccrine glands are found within the skin, and their function is to secrete water while conserving sodium chloride for electrolyte maintenance. The eccrine gland consists of a secretory coil and a duct.






